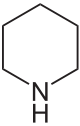
Piperidine
|
|
|||
 |
|||
| Names | |||
|---|---|---|---|
| IUPAC name
Piperidine
|
|||
| Other names
Hexahydropyridine
Azacyclohexane Pentamethyleneamine Azinane |
|||
| Identifiers | |||
| 110-89-4 |
|||
| ChEBI | CHEBI:18049 |
||
| ChEMBL | ChEMBL15487 |
||
| ChemSpider | 7791 |
||
| 5477 | |||
| Jmol 3D model | Interactive image | ||
| PubChem | 8082 | ||
| RTECS number | TM3500000 | ||
| UNII | 67I85E138Y |
||
|
|||
|
|||
| Properties | |||
| C5H11N | |||
| Molar mass | 85.15 g·mol−1 | ||
| Appearance | colourless liquid | ||
| Density | 0.862 g/mL, liquid | ||
| Melting point | −7 °C (19 °F; 266 K) | ||
| Boiling point | 106 °C (223 °F; 379 K) | ||
| miscible | |||
| Acidity (pKa) | 11.22[2] | ||
| Viscosity | 1.573 cP at 25 °C | ||
| Vapor pressure | {{{value}}} | ||
| Related compounds | |||
|
Related compounds
|
pyridine pyrrolidine piperazine |
||
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|||
| Infobox references | |||
Piperidine is an organic compound with the molecular formula (CH2)5NH. This heterocyclic amine consists of a six-membered ring containing five methylene bridges (-CH
2-) and one amine bridge (-NH-). It is a colorless fuming liquid with an odor described as ammoniacal or pepper-like;[3] the name comes from the genus name Piper, which is the Latin word for pepper.[4] Piperidine is a widely used building block and chemical reagent in the synthesis of organic compounds, including pharmaceuticals.
Contents
Production
Industrially, piperidine is produced by the hydrogenation of pyridine, usually over a molybdenum disulfide catalyst:[5]
- C5H5N + 3 H2 → C5H10NH
Pyridine can also be reduced to piperidine via a modified Birch reduction using sodium in ethanol.[6]
Natural occurrence of piperidine and derivatives
Piperidine itself has been obtained from black pepper,[7] from Psilocaulon absimile N.E.Br (Aizoaceae),[8] and in Petrosimonia monandra.[9]
The piperidine structural motif is present in numerous natural alkaloids. These include piperine, which gives black pepper its spicy taste. This gave the compound its name. Other examples are the fire ant toxin solenopsin,[10] the nicotine analog anabasine of the Tree Tobacco (Nicotiana glauca), lobeline of the Indian tobacco, and the toxic alkaloid coniine from poison hemlock, which was used to put Socrates to death.[11]
Conformation
Piperidine prefers a chair conformation, similar to cyclohexane. Unlike cyclohexane, piperidine has two distinguishable chair conformations: one with the N–H bond in an axial position, and the other in an equatorial position. After much controversy during the 1950s–1970s, the equatorial conformation was found to be more stable by 0.72 kcal/mol in the gas phase.[12] In nonpolar solvents, a range between 0.2 and 0.6 kcal/mol has been estimated, but in polar solvents the axial conformer may be more stable.[13] The two conformers interconvert rapidly through nitrogen inversion; the free energy activation barrier for this process, estimated at 6.1 kcal/mol, is substantially lower than the 10.4 kcal/mol for ring inversion.[14] In the case of N-methylpiperidine, the equatorial conformation is preferred by 3.16 kcal/mol,[12] which is much larger than the preference in methylcyclohexane, 1.74 kcal/mol.
 |
 |
|
|
|
Reactions
Piperidine is a widely used secondary amine. It is widely used to convert ketones to enamines.[15] Enamines derived from piperidine can be used in the Stork enamine alkylation reaction.[16]
Piperidine can be converted to the chloramine C5H10NCl with calcium hypochlorite. The resulting chloramine undergoes dehydrohalogenation to afford the cyclic imine.[17]
NMR chemical shifts
13C NMR = (CDCl3, ppm) 47.27.2, 25.2
1H NMR = (CDCl3, ppm) 2.79, 2.19, 1.51
Uses
Piperidine is used as a solvent and as a base. The same is true for certain derivatives: N-formylpiperidine is a polar aprotic solvent with better hydrocarbon solubility than other amide solvents, and 2,2,6,6-tetramethylpiperidine is a highly sterically hindered base, useful because of its low nucleophilicity and high solubility in organic solvents.
A significant industrial application of piperidine is for the production of dipiperidinyl dithiuram tetrasulfide, which is used as a rubber vulcanization accelerator.[5]
List of piperidine medications
Piperidine and its derivatives are ubiquitous building blocks in the synthesis of pharmaceuticals and fine chemicals. The piperidine structure is e.g. found in the pharmaceuticals:
- SSRI (Selective Serotonin Reuptake Inhibitors)
- Analeptics/Nootropics (Stimulants)
- SERM (Selective Estrogen Receptor Modulators)
- Vasodilators
- Neuroleptics (Antipsychotics)
- Opioids
- Pethidine (Meperidine)
- loperamide
- Other agents
- Psychochemical compounds
- melperone the psychochemical agents Ditran-B (JB-329), N-methyl-3-piperidyl benzilate (JB-336) and in many others.
Piperidine is also commonly used in chemical degradation reactions, such as the sequencing of DNA in the cleavage of particular modified nucleotides. Piperidine is also commonly used as a base for the deprotection of Fmoc-amino acids used in solid-phase peptide synthesis.
Piperidine is listed as a Table II precursor under the United Nations Convention Against Illicit Traffic in Narcotic Drugs and Psychotropic Substances due to its use (peaking in the 1970s) in the clandestine manufacture of PCP (also known as angel dust, sherms, wet, etc.).[18]
References
- ↑ International Chemical Safety Card 0317
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ 5.0 5.1 Karsten Eller, Erhard Henkes, Roland Rossbacher, Hartmut Höke "Amines, Aliphatic" Ullmann's Encyclopedia of Industrial Chemistry 2002 Wiley-VCH. doi:10.1002/14356007.a02_001
- ↑ Lua error in package.lua at line 80: module 'strict' not found.; Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Spaeth and Englaender, Ber.1935,68, 2218; cf. Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ 12.0 12.1 Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.; Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.; Lua error in package.lua at line 80: module 'strict' not found.
- ↑ List of Precursors and Chemicals Frequently Used in the Illicit Manufacture of Narcotic Drugs and Psychotropic Substances Under International Control, International Narcotics Control Board (link is dead)
Lua error in package.lua at line 80: module 'strict' not found.