Uranosena
senyawa kimia
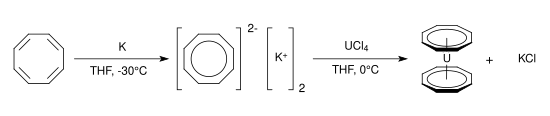
Uranocene, U(C8H8)2, adalah senyawa organouranium yang tersusun dari atom uranium yang diapit diantara dua cincin siklooktatetraenida. Senyawa tersebut adalah salah satu senyawa organoaktinida pertama yang disintesis. Senyawa tersebut adalah padatan hijau yang sensitif terhadap udara yang larut dalam pelarut organik. Uranosena, salah satu anggota dari "aktinosena," sekelompok metalosena yang menggabungkan unsur-unsur dari rangkaian aktinida.[1][2][3][4][5][6][7][8][9][10][11][12]
Referensi
sunting- ^ Seyferth, D. (2004). "Uranocene. The First Member of a New Class of Organometallic Derivatives of the f Elements". Organometallics. 23 (15): 3562–3583. doi:10.1021/om0400705 .
- ^ Zalkin, Allan; Raymond, Kenneth N. (1969). "Structure of di-.pi.-cyclooctatetraeneuranium (uranocene)". Journal of the American Chemical Society (dalam bahasa Inggris). 91 (20): 5667–5668. doi:10.1021/ja01048a055. ISSN 0002-7863.
- ^ Clark, J. P.; Green, J. C. (1977). "An Investigation of the Electronic Structure of Bis(eta-cyclo-octatetraene)-actinoids by Helium-(I) and -(II) Photoelectron Spectroscopy". J. Chem. Soc., Dalton Trans. (5): 505–508. doi:10.1039/DT9770000505.
- ^ Roesch, N.; Streitwieser, A. (1983). "Quasirelativistic SCF-Xalpha Scattered-Wave Study of Uranocene, Thorocene, and Cerocene". J. Am. Chem. Soc. 105 (25): 7237–7240. doi:10.1021/ja00363a004.
- ^ Chang, A. H. H.; Pitzer, R. M. (1989). "Electronic Structure and Spectra of Uranocene". J. Am. Chem. Soc. 111 (7): 2500–2507. doi:10.1021/ja00189a022.
- ^ Karraker, D. G.; Stone, J. A.; Jones, E. R.; Edelstein, N. (1970). "Bis(cyclooctatetraenyl)neptunium(IV) and Bis(cyclooctatetraenyl)plutonium(IV)". J. Chem. Phys. 92 (16): 4841–4845. doi:10.1021/ja00719a014.
- ^ Fischer, R. D. (1979). "NMR Spectroscopy of Organometallic Compounds of the f-Elements: Practical Applications". Dalam Marks, T. J.; Fischer, R. D. Volume 44 – Organometallics of the f-Elements. NATO Advanced Study Institutes Series: Series C – Mathematical and Physical Sciences. Dordrecht, Holland: Reidel. hlm. 337–377. ISBN 90-277-0990-4.
- ^ Hayes, R. G.; Edelstein, N. (1972). "An Elementary Molecular Orbital Calculation on U(C8H8)2 and Its Application to the Electronic Structure of U(C8H8)2, Np(C8H8)2. and Pu(C8H8)2". J. Am. Chem. Soc. 94 (25): 8688–8691. doi:10.1021/ja00780a008.
- ^ Liu, W.; Dolg, M.; Fulde, P. (1997). "Low-lying electronic states of lanthanocenes and actinocenes M(C8H8)2 (M=Nd, Tb, Yb, U)". J. Chem. Phys. 107 (9): 3584–3591. Bibcode:1997JChPh.107.3584L. doi:10.1063/1.474698.
- ^ Herzberg, G. (1966). Molecular Spectra and Molecular Structure III. Electronic Spectra and Electronic Structure of Polyatomic Molecules. Princeton, New Jersey: D. Van Nostrand. hlm. 566.
- ^ Dallinger, R. F.; Stein, P.; Spiro, T. G. (1978). "Resonance Raman Spectroscopy of Uranocene: Observation of an Anomalously Polarized Electronic Band and Assignment of Energy Levels". J. Am. Chem. Soc. 100 (25): 7865–7870. doi:10.1021/ja00493a013.
- ^ Hager, J. S.; Zahardis, J.; Pagni, R. M.; et al. (2004). "Raman under nitrogen. The high-resolution Raman spectroscopy of crystalline uranocene, thorocene, and ferrocene". J. Chem. Phys. 120 (6): 2708–2718. Bibcode:2004JChPh.120.2708H. doi:10.1063/1.1637586. PMID 15268415.
Wikimedia Commons memiliki media mengenai uranocene.
Bacaan lanjutan
sunting- The f elements, Nikolas Kaltsoyannis and Peter Scott. ISBN 0-19-850467-5
- Chemistry of the Elements, N. N. Greenwood and A. Earnshaw. ISBN 0-08-022057-6
- Lanthanides & Actinides: Organoactinides