Citronellal
From Infogalactic: the planetary knowledge core
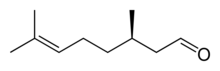 |
|
| Ball-and-stick model of the (+)-citronellal molecule (+)-Citronellal
|
|
| Ball-and-stick model of the (-)-citronellal molecule (-)-Citronellal
|
|
| Names | |
|---|---|
| IUPAC name
3,7-dimethyloct-6-en-1-al
|
|
| Identifiers | |
| 106-23-0 |
|
| ChEBI | CHEBI:47856 |
| ChEMBL | ChEMBL447944 |
| ChemSpider | 7506 |
| EC Number | 203-376-6 |
| Jmol 3D model | Interactive image Interactive image |
| KEGG | C17384 |
| PubChem | 7794 |
| UNII | QB99VZZ7GZ |
|
|
|
|
| Properties | |
| C10H18O | |
| Molar mass | 154.25 g/mol |
| Density | 0.855 g/cm3 |
| Boiling point | 201 to 207 °C (394 to 405 °F; 474 to 480 K) |
| Related compounds | |
|
Related alkenals
|
Citral |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
| Infobox references | |
Citronellal or rhodinal or 3,7-dimethyloct-6-en-1-al (C10H18O) is a monoterpenoid, the main component in the mixture of terpenoid chemical compounds that give citronella oil its distinctive lemon scent.
Citronellal is a major isolate in distilled oils from the plants Cymbopogon,[2] lemon-scented gum, and lemon-scented teatree. The (–)-(S)-enantiomer of citronellal makes up up to 80% of the leaf oil from kaffir lime leaves and is the compound responsible for its characteristic aroma.
Citronellal has insect repellent properties, and research shows high repellent effectiveness against mosquitoes.[3] Research shows that citronellal has strong antifungal qualities.[4]
Compendial status
Lua error in package.lua at line 80: module 'strict' not found.
See also
References
- ↑ Citronellal, The Merck Index, 12th Edition
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.