Hydrogen peroxide - urea
 |
|
 |
|
 |
|
| Names | |
|---|---|
| Other names
Urea peroxide, percarbamide, UHP
|
|
| Identifiers | |
| 124-43-6 |
|
| ChEBI | CHEBI:75178 |
| ChemSpider | 29034 |
| Jmol 3D model | Interactive image |
| PubChem | 31294 |
| UNII | 31PZ2VAU81 |
|
|
|
|
| Properties | |
| CH6N2O3 | |
| Molar mass | 94.07 g·mol−1 |
| Appearance | White solid |
| Melting point | 75 to 91.5 °C (167.0 to 196.7 °F; 348.1 to 364.6 K) (decomposes) |
| Pharmacology | |
| ATC code | D02 |
| Vapor pressure | {{{value}}} |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
| Infobox references | |
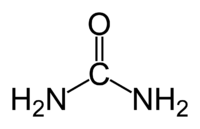
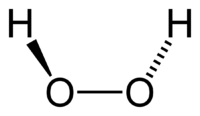
Hydrogen peroxide - urea is a solid composed of equal amounts of hydrogen peroxide and urea. This compound is a white crystalline solid, which dissolves in water to give free hydrogen peroxide. Often called carbamide peroxide in the dental applications, it is used as a source of hydrogen peroxide for bleaching, disinfection, and oxidation.
Contents
Structure, properties, production
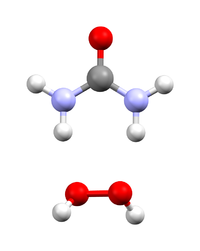
Akin to water of crystallization, hydrogen peroxide cocrystallizes with urea with the stoichiometry of 1:1. The compound is simply produced (on a scale of several hundred tonnes a year) by the dissolution of urea in excess concentrated hydrogen peroxide solution, followed by crystallization.[1] The laboratory synthesis is analogous.[2] Upon dissolving in various solvents, this 1:1 complex dissociates back to urea and hydrogen peroxide. So just like hydrogen peroxide, the adduct is an oxidizer. The solubility of commercial samples varies from 0.05 g/mL[3] to more than 0.6 g/mL.[4] The solid state structure of this adduct at the right has been determined by neutron diffraction.[5]
Applications
Disinfectant and bleaching agent
Hydrogen peroxide - urea is mainly used as a disinfecting and bleaching agent in cosmetics and pharmaceuticals.[1] As a drug, this compound is used in some preparations for the whitening of teeth.[1][6][7] It is also used to relieve minor inflammation of gums, oral mucosal surfaces and lips including canker sores and dental irritation,[8]and to emulsify and disperse ear wax.[9]
A pharmaceutical company, Elorac, has performed clinical trials for the use of carbamide peroxide for the treatment of acne vulgaris.[10]
Reagent in organic synthesis
In the laboratory, it is used as a more easily handled replacement for hydrogen peroxide.[2][11][12]
Safety
As an oxidizer, this compound is a skin, eye and respiratory irritant. At higher concentrations, it is corrosive and may cause burns if not used properly. If too much gel is used and left in contact with skin or gums, there is a risk of white chemical burns called blanching.[citation needed]
See also
References
- ↑ 1.0 1.1 1.2 Lua error in package.lua at line 80: module 'strict' not found.
- ↑ 2.0 2.1 Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Sigma-Aldrich specification sheet
- ↑ Chemicalland data sheet
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Toothwhitening from the UMD of New Jersey website
- ↑ Center for Integrative Medicine: Carbamide Peroxide from the University of Maryland Medical Center website Archived October 18, 2007 at the Wayback Machine
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Elorac Announces Clinical Results of Carbamide Peroxide Studies for the Treatment of Acne website
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Harry Heaney, Francesca Cardona, Andrea Goti, "Hydrogen Peroxide–Urea" Encyclopedia of Reagents for Organic Synthesis 2008. doi:10.1002/047084289X.rh047.pub2
External links
- Lua error in package.lua at line 80: module 'strict' not found.
- Lua error in package.lua at line 80: module 'strict' not found.