Petroleum naphtha
Petroleum naphtha is an intermediate hydrocarbon liquid stream derived from the refining of crude oil.[1][2][3] It is most usually desulfurized and then catalytically reformed, which re-arranges or re-structures the hydrocarbon molecules in the naphtha as well as breaking some of the molecules into smaller molecules to produce a high-octane component of gasoline (or petrol).
There are hundreds of different petroleum crude oil sources worldwide and each crude oil has its own unique composition or assay. There are also hundreds of petroleum refineries worldwide and each of them is designed to process either a specific crude oil or specific types of crude oils. Naphtha is a general term as each refinery produces its own naphthas with their own unique initial and final boiling points and other physical and compositional characteristics.
Naphthas may also be produced from other material such as coal tar, shale deposits, tar sands and the destructive distillation of wood.[4][5]
Contents
The major source of petroleum naphtha in a petroleum refinery
The first unit process in a petroleum refinery is the crude oil distillation unit. The overhead liquid distillate from that unit is called virgin or straight-run naphtha and that distillate is the largest source of naphtha in most petroleum refineries. The naphtha is a mixture of many different hydrocarbon compounds. It has an initial boiling point (IBP) of about 35 °C and a final boiling point (FBP) of about 200 °C, and it contains paraffins, naphthenes (cyclic paraffins) and aromatic hydrocarbons ranging from those containing 4 carbon atoms to those containing about 10 or 11 carbon atoms.
The virgin naphtha is often further distilled into two streams:[6]
- a virgin light naphtha with an IBP of about 30 °C and a FBP of about 145 °C containing most (but not all) of the hydrocarbons with 6 or less carbon atoms
- a virgin heavy naphtha containing most (but not all) of the hydrocarbons with more than 6 carbon atoms. The heavy naphtha has an IBP of about 140 °C and a FBP of about 205 °C.
It is the virgin heavy naphtha that is usually processed in a catalytic reformer because the light naphtha has molecules with 6 or less carbon atoms which, when reformed, tend to crack into butane and lower molecular weight hydrocarbons which are not useful as high-octane gasoline blending components. Also, the molecules with 6 carbon atoms tend to form aromatics which is undesirable because governmental environmental regulations in a number of countries limit the amount of aromatics (most particularly benzene) that gasoline may contain.[7][8][9]
Types of virgin naphthas
The table below lists some typical virgin heavy naphthas, available for catalytic reforming, derived from various crude oils. It can be seen that they differ significantly in their content of paraffins, naphthenes and aromatics:
Crude oil name 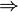 Location 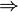 |
Barrow Island Australia[10] |
Mutineer-Exeter Australia[11] |
CPC Blend Kazakhstan[12] |
Draugen North Sea[13] |
|---|---|---|---|---|
| Initial boiling point, °C | 149 | 140 | 149 | 150 |
| Final boiling point, °C | 204 | 190 | 204 | 180 |
| Paraffins, liquid volume % | 46 | 62 | 57 | 38 |
| Naphthenes, liquid volume % | 42 | 32 | 27 | 45 |
| Aromatics, liquid volume % | 12 | 6 | 16 | 17 |
Cracked naphthas
Some refinery naphthas also contain some olefinic hydrocarbons, such as naphthas derived from the fluid catalytic cracking, visbreakers and coking processes used in many refineries. Those olefin-containing naphthas are often referred to as cracked naphthas.
In some (but not all) petroleum refineries, the cracked naphthas are desulfurized and catalytically reformed (as are the virgin naphthas) to produce additional high-octane gasoline components.
Other uses
Some petroleum refineries also produce small amounts of specialty naphthas for use as solvents, cleaning fluids, paint and varnish diluents, asphalt diluents, rubber industry solvents, dry-cleaning, cigarette lighters, and portable camping stove and lantern fuels. Those specialty naphthas are subjected to various purification processes.
Sometimes the specialty naphthas are called petroleum ether, petroleum spirits, mineral spirits, paraffin, benzine, hexanes, ligroin, white oil or white gas, painters naphtha, refined solvent naphtha and Varnish makers' & painters' naphtha (VM&P) . The best way to determine the boiling range and other compositional characteristics of any of the specialty naphthas is to read the Material Safety Data Sheet (MSDS) for the specific naphtha of interest.
On a much larger scale, petroleum naphtha is also used in the petrochemicals industry as feedstock to steam reformers and steam crackers for the production of hydrogen (which may be and is converted into ammonia for fertilizers), ethylene and other olefins. Natural gas is also used as feedstock to steam reformers and steam crackers.
Safety
People can be exposed to petroleum naphtha in the workplace by breathing it in, swallowing it, skin contact, and eye contact. The Occupational Safety and Health Administration (OSHA) has set the legal limit (permissible exposure limit) for petroleum naphtha exposure in the workplace as 500 ppm (2000 mg/m3) over an 8-hour workday. The National Institute for Occupational Safety and Health (NIOSH) has set a recommended exposure limit (REL) of 350 mg/m3 over an 8-hour workday and 1800 mg/m3 over 15 minutes. At levels of 1100 ppm, 10% of the lower explosive limit, petroleum naphtha is immediately dangerous to life and health.[14]
References
- This article incorporates material from the Citizendium article "Petroleum naphtha", which is licensed under the Creative Commons Attribution-ShareAlike 3.0 Unported License but not under the GFDL.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Exploiting the Benefits of Fischer-Tropsch Technology (Sasol’s integrated business model)
- ↑ Beychok, M.R., Process and environmental technology for producing SNG and liquid fuels, U.S. EPA report EPA-660/2-75-011, May 1975
- ↑ Fuel Chemistry (scroll down to "What is naphtha")
- ↑ Canadian regulations on benzene in gasoline
- ↑ Briefing on Benzene in Petrol From website of United Kingdom Petroleum Industry Association (UKPIA)
- ↑ USA regulations on benzene in gasoline
- ↑ Barrow Island crude oil assay
- ↑ Mutineer-Exeter crude oil assay
- ↑ CPC Blend crude oil assay
- ↑ Draugen crude oil assay
- ↑ Lua error in package.lua at line 80: module 'strict' not found.