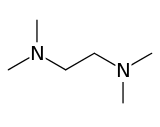
Tetramethylethylenediamine
 |
|
 |
|
| Names | |
|---|---|
| Other names
N,N,N′,N′-Tetramethylethane-1,2-diamine[1]
|
|
| Identifiers | |
| 110-18-9 |
|
| Abbreviations | TMEDA, TEMED |
| 1732991 | |
| ChEBI | CHEBI:32850 |
| ChemSpider | 7746 |
| EC Number | 203-744-6 |
| 2707 | |
| Jmol 3D model | Interactive image |
| MeSH | N,N,N',N'-tetramethylethylenediamine |
| PubChem | 8037 |
| RTECS number | KV7175000 |
| UN number | 2372 |
|
|
|
|
| Properties | |
| C6H16N2 | |
| Molar mass | 116.21 g·mol−1 |
| Appearance | Colorless liquid |
| Odor | Fishy, ammoniacal |
| Density | 0.7765 g mL−1 (at 20 °C) |
| Melting point | −58.6 °C; −73.6 °F; 214.5 K |
| Boiling point | 121.1 °C; 249.9 °F; 394.2 K |
| Miscible | |
| Acidity (pKa) | 8.97 |
| Basicity (pKb) | 5.85 |
|
Refractive index (nD)
|
1.4179 |
| Vapor pressure | {{{value}}} |
| Related compounds | |
|
Related amines
|
Triethylenetetramine |
|
Related compounds
|
|
| Supplementary data page | |
| Refractive index (n), Dielectric constant (εr), etc. |
|
|
Thermodynamic
data |
Phase behaviour solid–liquid–gas |
| UV, IR, NMR, MS | |
| Infobox references | |
Tetramethylethylenediamine (TMEDA or TEMED) is a chemical compound with the formula (CH3)2NCH2CH2N(CH3)2. This species is derived from ethylenediamine by replacement of the four N-H groups with four N-methyl groups. It is a colorless liquid, although old samples often appear yellow. Its odor is remarkably similar to that of rotting fish.[2]
As a reagent in organic and inorganic synthesis
TMEDA is widely employed as a ligand for metal ions. It forms stable complexes with many metal halides, e.g. zinc chloride and copper(I) iodide, giving complexes that are soluble in organic solvents. In such complexes, TMEDA serves as a bidentate ligand.
TMEDA has an affinity for lithium ions.[2] When mixed with n-butyllithium, TMEDA's nitrogen atoms coordinate to the lithium, forming a cluster of higher reactivity than the tetramer or hexamer that n-butyllithium normally adopts. BuLi/TMEDA is able to metallate or even doubly metallate many substrates including benzene, furan, thiophene, N-alkylpyrroles, and ferrocene.[2] Many anionic organometallic complexes have been isolated as their [Li(tmeda)2]+ complexes.[3] In such complexes [Li(tmeda)2]+ behaves like a quaternary ammonium salt, such as [NEt4]+.
It is also worth noting that sBuLi/TMEDA is also a useful combination in organic synthesis. Utilization of this is useful in cases where the n-butyl anion is able to add into the starting material due to its weak nucleophilic nature. TMEDA is still capable of forming a metal complex with Li in this case as mentioned above.
Other uses
TMEDA is used with ammonium persulfate to catalyze the polymerization of acrylamide when making polyacrylamide gels, used in gel electrophoresis, for the separation of proteins or nucleic acids. Although the amounts used in this technique may vary from method to method, 0.1-0.2% v/v TMEDA is a "traditional" range. TMEDA can also be a component of Hypergolic propellants.
References
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ 2.0 2.1 2.2 Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ K. W. Henderson, A. E. Dorigo, Qi-Long Liu, P. G. Williard "Effect of Polydentate Donor Molecules on Lithium Hexamethyldisilazide Aggregation: An X-ray Crystallographic and a Combination Semiempirical PM3/Single Point ab Initio Theoretical Study" J. Am. Chem. Soc. 1997, volume 119, pp. 11855. doi:10.1021/ja971920t
