Sodium perchlorate
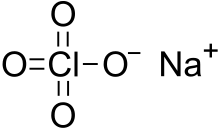
| |

| |
| Names | |
|---|---|
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.028.647 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1502 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| NaClO4 (anhydrous) NaClO4·H2O (monohydrate) | |
| Molar mass | 122.44 g/mol (anhydrous) 140.45 g/mol (monohydrate) |
| Appearance | White crystalline solid |
| Density | 2.4994 g/cm3 (anhydrous) 2.02 g/cm3 (monohydrate) |
| Melting point | 468 °C (874 °F; 741 K) (decomposes, anhydrous) 130 °C (monohydrate) |
| Boiling point | 482 °C (900 °F; 755 K) (decomposes, monohydrate) |
| 209.6 g/(100 mL) (25 °C, anhydrous) 209 g/(100 mL) (15 °C, monohydrate) | |
Refractive index (nD)
|
1.4617 |
| Structure | |
| orthorhombic | |
| Hazards | |
| GHS labelling: | |
  
| |
| Danger | |
| H271, H302, H319, H373 | |
| P102, P220, P305+P351+P338, P338 | |
| NFPA 704 (fire diamond) | |
| Flash point | 400 °C (752 °F; 673 K) |
| Safety data sheet (SDS) | ICSC 0715 |
| Related compounds | |
Other anions
|
|
Other cations
|
|
Related compounds
|
Perchloric acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sodium perchlorate is an inorganic compound with the chemical formula NaClO4. It consists of sodium cations Na+ and perchlorate anions ClO−4. It is a white crystalline, hygroscopic solid that is highly soluble in water and ethanol. It is usually encountered as sodium perchlorate monohydrate NaClO4·H2O. The compound is noteworthy as the most water-soluble of the common perchlorate salts.
Sodium perchlorate and other perchlorates has been found on the planet Mars, first detected by the NASA probe Phoenix in 2009. This was later confirmed by spectral analysis by the Mars Reconnaissance Orbiter in 2015 of what is thought to be brine seeps which may be the first evidence of flowing liquid water containing hydrated salts on Mars.[1][2]
Selected properties
[edit]Its heat of formation is −382.75 kJ/mol, i.e. it is thermally stable up to high temperatures. At 490 °C it undergoes thermal decomposition, producing sodium chloride and dioxygen.[3] It crystallizes in the rhombic crystal system.[4]
Uses
[edit]Perchloric acid is made by treating NaClO4 with HCl.[5] Ammonium perchlorate and potassium perchlorate, of interest in rocketry and pyrotechnics, are prepared by double decomposition from a solution of sodium perchlorate and ammonium chloride or potassium chloride, respectively.
Laboratory applications
[edit]Because of its high solubility (2096 g/L at 25 °C) and the inert behaviour of dissolved perchlorate, solutions of NaClO4 are often used as unreactive background electrolyte (supporting electrolyte). Indeed, because the reduction reaction of perchlorate is kinetically limited even if it is a thermodynamically unstable compound, perchlorate is a redox non-sensitive anion. It is also a non-complexing anion with no ligand binding capacity.
Sodium perchlorate is the precursor to ammonium, potassium and lithium perchlorate salts, often taking advantage of their low solubility relative to NaClO4 (209 g/(100 mL) at 25 °C).[6]
It is used for denaturating proteins in biochemistry and in standard DNA extraction and hybridization reactions in molecular biology.
In medicine
[edit]Sodium perchlorate can be used to block iodine uptake before administration of iodinated contrast agents in patients with subclinical hyperthyroidism (suppressed TSH).[7]
Production
[edit]Sodium perchlorate is produced by anodic oxidation of sodium chlorate (NaClO3) at an inert electrode, such as platinum.[5]
- Na+ClO−3 + H2O → Na+ClO−4 + 2 H+ + 2 e− (acidic medium)
- Na+ClO−3 + 2 OH− → Na+ClO−4 + H2O + 2 e− (alkaline medium)
Safety
[edit]Acute toxicity: The median lethal dose (LD50) is 2 – 4 g/kg (rabbits, oral).[5]
Chronic toxicity: The frequent consumption of drinking water with low concentrations (in the range of μg/L, ppb) of perchlorate is harmful for the thyroid gland as the perchlorate anion competes with the uptake of iodide.
See also
[edit]References
[edit]- ^ Wadsworth, Jennifer; Cockell, Charles S. (July 6, 2017). "Perchlorates on Mars enhance the bacteriocidal effects of UV light". Scientific Reports. 7 (2017, #4662): 4662. Bibcode:2017NatSR...7.4662W. doi:10.1038/s41598-017-04910-3. PMC 5500590. PMID 28684729.
- ^ Delbecq, Denis (September 28, 2015). "De l'eau liquide répérée sur les pentes martiennes" [Liquid water spotted on Martian slopes]. Le Temps (in French). Archived from the original on January 18, 2017. Retrieved August 23, 2021.
- ^ Devlin, D. J.; Herley, P. J. (1987). "Thermal decomposition and dehydration of sodium perchlorate monohydrate". Reactivity of Solids. 3 (1–2): 75–84. doi:10.1016/0168-7336(87)80019- (inactive 2024-09-12). Retrieved 3 May 2023.
{{cite journal}}: CS1 maint: DOI inactive as of September 2024 (link) - ^ Eagleson, Mary (1994). Concise Encyclopedia Chemistry. revised, illustrated. Walter de Gruyter. p. 1000. ISBN 978-3-11-011451-5. Retrieved March 7, 2013.
- ^ a b c Helmut Vogt; Jan Balej; John E. Bennett; Peter Wintzer; Saeed Akbar Sheikh; Patrizio Gallone (2000). "Chlorine Oxides and Chlorine Oxygen Acids". Ullmann's Encyclopedia of Industrial Chemistry. Wiley-VCH. doi:10.1002/14356007.a06_483. ISBN 3-527-30673-0.
- ^ Angus, Patricia M.; Jackson, W. Gregory (1994). "Linkage Isomerism in Cobalt(III) Pentaammine Complexes of 2-Pyridone". Inorganic Chemistry. 33 (3): 477–483. doi:10.1021/ic00081a014.
- ^ Becker, C. (2007, September). Radiologisch praxisrelevante prophylaxe und therapie von nebenwirkungen jodhaltiger kontrastmittel. [Prophylaxis and treatment of side effects due to iodinated contrast media relevant to radiological practice]. Der Radiologe. 47(9), 768–773. doi:10.1007/s00117-007-1550-4
