Dalcetrapib
 |
|
| Names | |
|---|---|
| IUPAC name
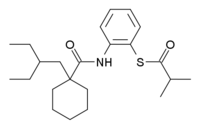
S-[2-({[1-(2-ethylbutyl)cyclohexyl]carbonyl}amino)phenyl] 2-methylpropanethioate
|
|
| Identifiers | |
| 211513-37-0 | |
| ChEMBL | ChEMBL313006 |
| ChemSpider | 5293737 |
| Jmol 3D model | Interactive image Interactive image |
| PubChem | 6918540 |
| UNII | 3D050LIQ3H |
|
|
|
|
| Properties | |
| C23H35NO2S | |
| Molar mass | 389.5945 |
| Vapor pressure | {{{value}}} |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
| Infobox references | |
Dalcetrapib or JTT-705 is a CETP inhibitor which was being developed by Hoffmann–La Roche until May 2012.[1][2] The drug was aimed at raising the blood levels of "good cholesterol" (cholesterol carried in HDL particles, aka HDL-C).[3] Prevailing observations indicate that high HDL levels correlate with better overall cardiovascular health, though it remains unclear whether raising HDL levels consequently leads to an increase in cardiovascular health.[4]
A 24-week clinical trial showed that dalcetrapib did increase HDL-C levels, supporting the agent's desired effect.[5] Further, the dal-PLAQUE phase IIb trial found evidence of plaque reduction.[6] Plaque reduction is an anticipated observation following an increase in HDL.[citation needed]
As of 2010[update] five phase II trials had started and there was no evidence of the raised blood pressure seen with torcetrapib.[5]
dal-VESSEL phase IIb trial found no evidence of flow-mediated dilatation improvement. A 17% increase of Lp-PLA2 mass level was noted.[7] Lp-PLA2 is associated with coronary heart disease and stroke.[citation needed]
dal-OUTCOMES phase III trial passed its first interim review in July, 2011,[8] however, development was halted on May 7, 2012 “due to a lack of clinically meaningful efficacy.”.[2]
The results of dal-OUTCOMES III were published in November, 2012.[9]
See also
- CETP inhibitor, which contains links to related agents; as of November 2012: Torcetrapib, Anacetrapib, Dalcetrapib and Evacetrapib
References
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ 2.0 2.1 Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ 5.0 5.1 Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.