Nabilone
Lua error in package.lua at line 80: module 'strict' not found.
 |
|
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
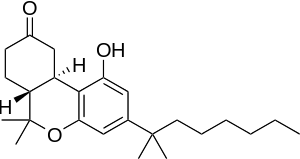
(6aR,10aR)-rel-1-hydroxy-6,6-dimethyl-3-(2-methyloctan-2-yl)-
|
|
| Clinical data | |
| Trade names | Cesamet, Canemes |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a607048 |
| Pregnancy category |
|
| Legal status |
|
| Routes of administration |
Oral form (PO)- capsule |
| Pharmacokinetic data | |
| Bioavailability | 20% after first-pass by the liver |
| Protein binding | similar to THC (+/-97%) |
| Biological half-life | 2 hours, with metabolites around 35 hours. |
| Identifiers | |
| CAS Number | 51022-71-0 |
| ATC code | A04AD11 (WHO) |
| PubChem | CID: 5284592 |
| DrugBank | DB00486 |
| ChemSpider | 4447641 |
| UNII | 2N4O9L084N |
| KEGG | D05099 |
| ChEMBL | CHEMBL947 |
| Chemical data | |
| Formula | C24H36O3 |
| Molecular mass | 372.541 g/mol |
|
|
|
|
| (verify) | |
Nabilone is a synthetic cannabinoid with therapeutic use as an antiemetic and as an adjunct analgesic for neuropathic pain. It mimics tetrahydrocannabinol (THC), the primary psychoactive compound found naturally occurring in Cannabis.[1]
In Canada, the United States, the United Kingdom and Mexico, nabilone is marketed as Cesamet. It was approved in 1985 by the U.S. Food and Drug Administration (FDA) for treatment of chemotherapy-induced nausea and vomiting (CINV) that has not responded to conventional antiemetics. Though it was approved by the FDA in 1985, the drug only began marketing in the United States in 2006. In Austria Nabilone is marketed as Canemes and got its approval for CINV in 2013.[2]
Although it doesn't have any indication officially (except in Mexico), nabilone is widely used as an adjunct therapy for chronic pain management. Numerous trials and case studies have demonstrated modest effectiveness for relieving fibromyalgia[3] and multiple sclerosis.[4]
Nabilone is a racemic mixture consisting of the (S,S) and the (R,R) isomers ("trans").
Medical uses
Nabilone has shown modest effectiveness in relieving fibromyalgia.[3] A 2011 systematic review of cannabinoids for chronic pain determined there was evidence of safety and modest efficacy for some conditions.[5]
The main settings that have seen published clinical trials of nabilone include movement disorders such as parkinsonism, chronic pain, dystonia and spasticity neurological disorders, multiple sclerosis, and the nausea of cancer chemotherapy. Nabilone is also effective in the treatment of inflammatory bowel disease, especially ulcerative colitis. Medical cannabis patients report that nabilone is more similar in effect to cannabidiol (CBD) than tetrahydrocannabinol (THC), indicating that it has more of a therapeutic effect on the body than a "high" effect on the mind.[citation needed]
A study comparing nabilone with metoclopramide, conducted before the development of modern 5-HT3 antagonist anti-emetics such as ondansetron, revealed that patients taking cisplatin chemotherapy preferred metoclopramide, while patients taking carboplatin preferred nabilone to control nausea and vomiting.[6]
Nabilone is also occasionally used for the adjuvant treatment of severe anxiety.[medical citation needed]
Research
Nabilone is sometimes used for nightmares in PTSD, but there have not been studies longer than nine weeks, so effects of longer term use are not known.[7] Nabilone has also been used for medication overuse headache.[8]
Pharmacokinetics
Nabilone is given in 1 or 2 mg doses multiple times a day up to a total of 6 mg. It is completely absorbed from oral administration and highly plasma protein bound. Multiple P450 enzymes extensively metabolize nabilone to various metabolites that have not been fully characterized.[9]
Adverse effects
Nabilone can increase, rather than decrease, post-operative pain; in the treatment of fibromyalgia, adverse effects limits the useful dose.[3] Adverse effects of nabilone include, but are not limited to dizziness/vertigo, euphoria, drowsiness, dry mouth, ataxia, sleep disturbance, dysphoria, headache, nausea, disorientation, depersonalization, asthenia [9]
See also
References
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ 3.0 3.1 3.2 Lua error in package.lua at line 80: module 'strict' not found.
- ↑ [non-primary source needed]Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ [non-primary source needed]Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ Lua error in package.lua at line 80: module 'strict' not found.
- ↑ 9.0 9.1 Lua error in package.lua at line 80: module 'strict' not found.
- Wikipedia articles needing factual verification from January 2014
- Drugs with non-standard legal status
- Chemical articles having calculated molecular weight overwritten
- Infobox drug articles without a structure image
- Articles with unsourced statements from April 2013
- Articles with unsourced statements from August 2015
- Benzochromenes
- Cannabinoids
- Ketones
- Phenols